Home

About
Virginia-Maryland College of Veterinary Medicine has a robust research program that spans basic biomedical sciences in infectious (viral, bacterial, and parasitic) and non-infectious (immune-mediated and oncologic) diseases to veterinary clinical research to translational to public health. The program's overall goal is to apply our research to improve the lives of pets and people.
Basic biomedical sciences include the use of appropriate animal models for diseases to better understand the disease process, a prerequisite for the development of better preventive and therapeutic strategies.
With a focus on One Health, our program brings together veterinarians, physicians, and other scientists to address public health threats affecting both people and animals.
Our translational research approach aims to take laboratory findings directly to clients in a clinical setting.
Ongoing clinical trials in our hospitals test research discoveries to develop better methods of disease prevention, diagnosis, and treatment.
Our faculty, students, staff, and research partners are committed to discovery, service, and training future researchers and veterinarians.
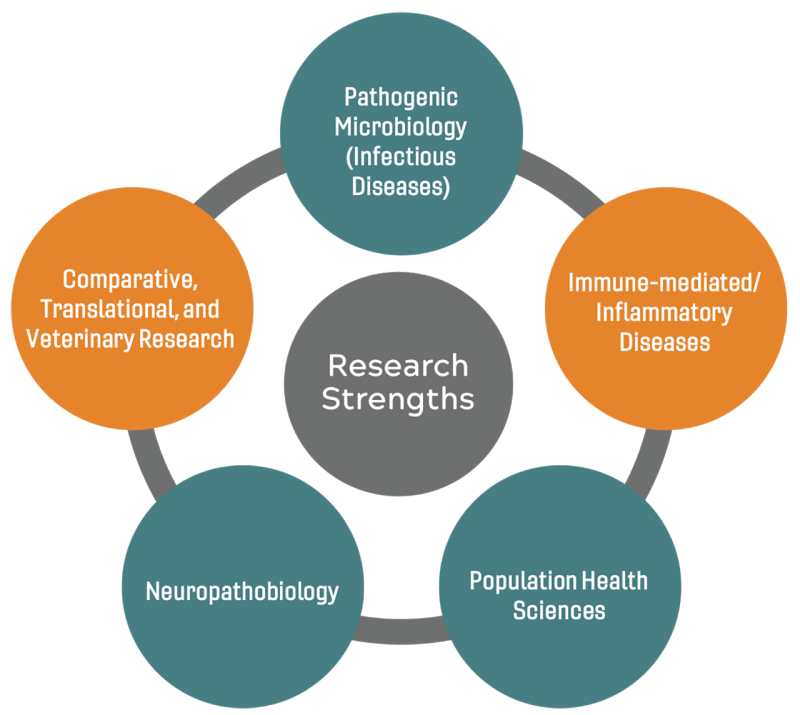
Signature Research Programs
-
Article Item
 Center for One Health Research , article
Center for One Health Research , articleThe Center for One Health Research (COHR) gathers faculty, staff, and students from VA-MD Vet Med and the Edward Via College of Osteopathic Medicine who have a shared interest in cooperative research and scholarship related to infectious diseases. This collaboration is founded on the two schools’ One Health philosophy, which is based on the understanding that animal health, human health, and the environment are inextricably linked; consequently, professionals from these fields must work together to protect, promote, and improve overall health. The center coordinates research, instruction, and outreach activities related to understanding the disease processes that affect both animal and human health, and supports the continued advancement of the science and technology of disease-intervention strategies.
-
Article Item
 Comparative, Translational, and Veterinary Research , article
Comparative, Translational, and Veterinary Research , articleResearch themes include clinical veterinary medicine research and translational medicine from laboratory animals to clinical veterinary and human patients; stem cell biology and its application; clinical trials management; and human-animal bond/interactions.
-
Article Item
 Immune-mediated / Inflammatory Diseases Research , article
Immune-mediated / Inflammatory Diseases Research , articleResearch themes include animal models for immune-mediated diseases, and understanding the mechanisms of such as autoimmune diseases and inflammatory diseases (eg. inflammatory bowel disease). Specific interests include role of epigenetics, microbiome, and environmental hormonal factors on these diseases; transition of inflammation leading to cancer; induction of immunity in oncologic conditions; and translational research.
-
Article Item
 Neuropathobiology Research , article
Neuropathobiology Research , articleResearch themes include animal models for neurodevelopment and traumatic injury, neurovascularization, neuroinflammation, and neuro-oncology; and translational research.
-
Article Item
 Pathogenic Microbiology / Infectious Diseases Research , article
Pathogenic Microbiology / Infectious Diseases Research , articleResearch themes include animal models for human and veterinary infectious diseases; and understanding the pathogenesis of the diseases, immunity to infectious and its prevention: development of vaccines and drugs, mechanisms of drug resistance, and translational research.
-
Article Item
 Public Health Research , article
Public Health Research , articleResearch focused on infectious diseases epidemiology and public health education that addresses the individual, social, and ecological determinants of health, reflects the human-animal-environment One Health interface, promotes health equity, and builds healthy communities.
Laboratories
-
Article Item
-
Article Item
-
Article Item
Research Training Programs
Latest News
-
Article Item
 Virginia Tech researchers work to make yoga accessible to everyone , article
Virginia Tech researchers work to make yoga accessible to everyone , articleThe project, a collaboration between the College of Agriculture and Life Sciences, Virginia Cooperative Extension, and the University Libraries, aims to reach historically underrepresented populations in yoga.
Date: Apr 02, 2024 -
Article Item
 Veterinary college symposium showcases graduate students’ research, makes collaboration connections , article
Veterinary college symposium showcases graduate students’ research, makes collaboration connections , articleMore than 60 graduate students from the veterinary college and its research and public health programs created posters explaining their research with some of them chosen to give either a full-length or four-minute flash talk about their research to their peers and faculty mentors.
Date: Mar 28, 2024 -
Article Item
 'Curious Conversations' podcast: Audrey Ruple talks about the Dog Aging Project , article
'Curious Conversations' podcast: Audrey Ruple talks about the Dog Aging Project , articleRuple discussed the Dog Aging Project, the largest-known study of dog health that aims to understand the keys to healthy aging in dogs and the risks to their health. She explained what information they are collecting, what it means for dogs, and how it might also be used to better understand human health.
Date: Mar 25, 2024 -
Article Item
 Stanton Foundation grant to fund canine mitral valve diagnosis research at veterinary college , article
Stanton Foundation grant to fund canine mitral valve diagnosis research at veterinary college , articleResearch will determine if veterinarians using only a stethoscope and chest X-rays can be effective in identifying dogs that would benefit from medical treatment for mitral valve disease.
Date: Mar 21, 2024 -
Article Item
 Two Fralin Biomedical Research Institute postdoctoral associates awarded American Heart Association fellowships , article
Two Fralin Biomedical Research Institute postdoctoral associates awarded American Heart Association fellowships , articleThe awards will help Karthi Sreedevi and Samar Antar advance their academic careers while conducting research related to cardiovascular disease.
Date: Mar 19, 2024 -
Article Item
 New research aims to use AI to make cancer diagnostics for pets more available and affordable , article
New research aims to use AI to make cancer diagnostics for pets more available and affordable , articlePh.D. student Christina Pacholec will use artificial intelligence to analyze thousands of cytological images in an effort to identify lymphoma in dogs more quickly and less invasively.
Date: Mar 12, 2024 -
Article Item
 Rare Disease Day brings worldwide attention to more than 7,000 rare diseases , article
Rare Disease Day brings worldwide attention to more than 7,000 rare diseases , articleVirginia Tech research into rare diseases — those that individually affect 200,000 or fewer but collectively touch 1 in 10 people in the U.S. — provides hope in identifying, treating, and supporting patients.
Date: Feb 28, 2024 -
Article Item
 Audrey Ruple collaborates with Dog Aging Project, largest-known study of dog health , article
Audrey Ruple collaborates with Dog Aging Project, largest-known study of dog health , articleInformation collected on more than 47,000 dogs is helping scientists explore many angles of animal, environmental, and human health.
Date: Feb 28, 2024